A ratiometric hypochlorite sensor guided by PET controlled ESIPT output with real time application in commercial bleach†‡
Abstract
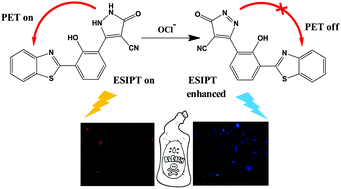
A HydroxyBenzothiazolyl dihydroPyrazole conjugate (HBP) featuring a modified acetylphenylhydrazine species armed with an N–N single bond forbidden from rotating was designed and employed for the sensitive and selective detection of hypochlorite (OCl−) anion. The probe exhibited weak fluorescence on account of photoinduced electron transfer (PET) from hydroxyphenyl dihydropyrazole moiety to benzothiazole which promptly changed upon OCl− induced oxidation of the dihydropyrazole species to pyrazole enhancing excited state intramolecular proton transfer (ESIPT) in the hydroxybenzothiazole counterpart. This led to excellent ratiometric fluorescence changes in the probe that conclusively proved its effectiveness at sensing OCl−. This unique, specific and fast oxidation was achieved selectively by OCl− as evidenced by competition experiments with various other reactive oxygen species (ROS), reactive nitrogen species (RNS) and anions. The mechanism has been supported by 1H-NMR titration studies along with DFT calculations. Notably, the probe responded remarkably to hypochlorite present in commercial bleach. Finally, the probe proved amenable to OCl− sensing in living matrices with no interference from trace amounts of ROS and RNS that inhabit living cells.


 Please wait while we load your content...
Please wait while we load your content...