Controllable synthesis and photoreduction performance towards Cr(vi) of BiOCl microrods with exposed (110) crystal facets†
Abstract
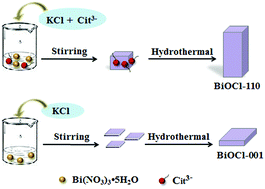
Bismuth oxyhalides have been of great interest, since they induce a strong internal static electric field and promote the effective separation of photoinduced electron–hole pairs, which enables good photocatalytic performance. However, simple synthesis and easily controllable facet exposure remain great challenges in this regard. Here, BiOCl microrods were synthesized via a simple hydrothermal method, using sodium citrate as a capping agent. High-resolution transmission electron microscopy (HRTEM) images show that the BiOCl microrods have (110) facets (BiOCl-110) exposed and grow with a [1![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 2] orientation. X-ray photoelectron spectroscopy (XPS) and FT-IR spectra show a number of citrate ions on the surface of BiOCl-110. An Ostwald ripening growth mechanism for BiOCl-110 has been demonstrated in our study. In addition, the photocatalytic performance of BiOCl-110 was evaluated via the degradation of hexavalent chromium (Cr(VI)) under solar light irradiation. From this, BiOCl-110 exhibits more excellent photoreduction performance towards Cr(VI) than BiOCl-001, and Cr(VI) at a concentration of 30 mg L−1 can be completely photoreduced within 4 and 10 min under acidic and neutral conditions, respectively. This enhanced photoreduction performance is ascribed to the more negative conductive band position of the (110) crystal facets, which enhances the reductive ability of photoelectrons.
2] orientation. X-ray photoelectron spectroscopy (XPS) and FT-IR spectra show a number of citrate ions on the surface of BiOCl-110. An Ostwald ripening growth mechanism for BiOCl-110 has been demonstrated in our study. In addition, the photocatalytic performance of BiOCl-110 was evaluated via the degradation of hexavalent chromium (Cr(VI)) under solar light irradiation. From this, BiOCl-110 exhibits more excellent photoreduction performance towards Cr(VI) than BiOCl-001, and Cr(VI) at a concentration of 30 mg L−1 can be completely photoreduced within 4 and 10 min under acidic and neutral conditions, respectively. This enhanced photoreduction performance is ascribed to the more negative conductive band position of the (110) crystal facets, which enhances the reductive ability of photoelectrons.


 Please wait while we load your content...
Please wait while we load your content...