A new multi-analyte fluorogenic sensor for efficient detection of Al3+ and Zn2+ ions based on ESIPT and CHEF features†
Abstract
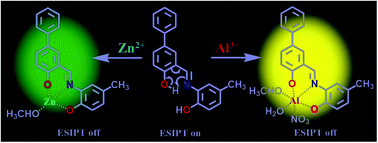
The fluorogenic chemosensor 3-(((2-hydroxy-4-methylphenyl)imino)methyl)-[1,1′-biphenyl]-4-ol (H2L) efficiently detects Zn2+ and Al3+ ions and subsequently fluoride ion in methanol–water (4/1, v/v, pH = 7.2) solution. The probe itself is non-emissive but upon treatment with Al3+ and Zn2+, it exhibits high fluorescence emission at two different wavelengths of 546 nm and 529 nm, respectively. Both excited-state intramolecular proton transfer (ESIPT) and chelation enhanced fluorescence (CHEF) processes play important roles in the enhancement of fluorescence intensity. Chelation of Zn2+ and Al3+ with the probe (H2L) inhibits C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and ESIPT which consequently enhances the emission intensity. The emission intensity of H2L–Al3+ is selectively quenched upon titration with F− anions. The structure of the probe is confirmed by the single crystal X-ray diffraction method. The electronic structure and sensing mechanism of the probe (H2L) are supported by density functional theory (DFT) and time-dependent density functional theory (TDDFT).
N isomerization and ESIPT which consequently enhances the emission intensity. The emission intensity of H2L–Al3+ is selectively quenched upon titration with F− anions. The structure of the probe is confirmed by the single crystal X-ray diffraction method. The electronic structure and sensing mechanism of the probe (H2L) are supported by density functional theory (DFT) and time-dependent density functional theory (TDDFT).


 Please wait while we load your content...
Please wait while we load your content...