The effects of protonated heterocyclic cations on the structural and magnetic properties of tetrachlorocuprate(ii) anions; X-ray, magnetochemical and EPR studies†
Abstract
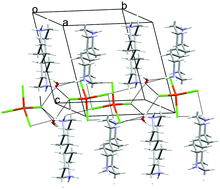
The crystal and molecular structures and magnetic properties of two new complexes (HMepy)2[CuCl4]·2H2O, 2, and (H2Me2ppz)[CuCl4], 3, formed by tetrachlorocuprate(II) anions and organic heterocyclic cations, bis(3-amino-2-chloro-5-methylpyridinium) (HMepy) and trans-2,5-dimethylpiperazinium (H2Me2ppz), respectively, have been determined by the analysis of their X-ray diffraction data, magnetic susceptibility behaviour and EPR spectral parameters. These characteristics differentiate the [CuCl4]2− anions in 2 and 3 as well as that previously reported in (H2bipip)[CuCl4]·H2O, 1, (bipip = 4,4′-bipiperidine) (Tuikka et al., CrystEngComm, 2013, 15, 6177; Wikaira et al., J. Coord. Chem., 2016, 69(1), 57). The present study reveals a significant effect of the organic cations on the geometry and magnetic properties of the [CuCl4]2− anions caused evidently by the changes in the crystal packing structure. A characteristic relationship was observed between the average cis-angles around the Cu(II) ions in [CuCl4]2− for 1–3, affected by the Cu(II) coordination geometry, and g-tensor components derived from the EPR spectra, a measure of interaction of copper(II) unpaired electrons in the [CuCl4]2− anions with an external magnetic field. The magneto-structural correlations revealed weak antiferromagnetic exchange interactions between the [CuCl4]2− anions transmitted by different pathways.


 Please wait while we load your content...
Please wait while we load your content...