A green and facile method to prepare graphitic carbon nitride nanosheets with outstanding photocatalytic H2O2 production ability via NaClO hydrothermal treatment
Abstract
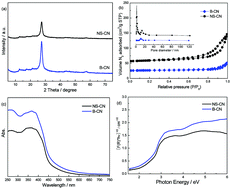
Transportation and transformation of photogenerated carriers during the photocatalytic process of graphitic carbon nitride (g-C3N4) are restricted by the low surface area and insufficient charge separation efficiency. Exfoliation of g-C3N4 into few layered nanosheets is an ideal method to resolve these problems. This work provides a green and facile method to prepare g-C3N4 nanosheets with outstanding photocatalytic H2O2 production ability via NaClO hydrothermal treatment. This simple process could readily be extended to a kilogram scale. XRD, UV-vis, N2 adsorption, SEM, TEM, XPS, O2-TPD, EIS and PL were used to characterize the catalysts. Taking advantages of both the oxidation effect of ClO− and the intercalation effect of Na+, the obtained g-C3N4 nanosheets display a specific surface area as high as 145.6 m2 g−1. The carbon vacancies formed by NaClO treatment could promote the O2 adsorption capacity of the catalyst and then activate the adsorbed O2 molecule. The charge separation efficiency and redox ability of g-C3N4 nanosheets are also promoted compared with bulk g-C3N4, leading to the obviously improved H2O2 production ability. In addition, the obtained g-C3N4 nanosheets show higher photocatalytic performance than that prepared by chemical oxidation exfoliation, thermal exfoliation and intercalation exfoliation methods. This confirms that NaClO hydrothermal treatment is a more green, facile and effective method to prepare g-C3N4 nanosheet photocatalysts with outstanding performance.


 Please wait while we load your content...
Please wait while we load your content...