Chemoenzymatic synthesis of a cholesterol-g-poly(amine-co-ester) carrier for p53 gene delivery to inhibit the proliferation and migration of tumor cells†
Abstract
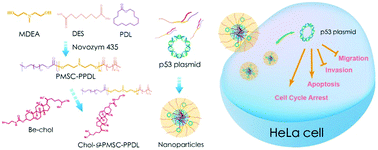
An amphiphilic cholesterol-g-poly(amine-co-ester) synthesized via a chemoenzymatic route has been successfully applied as a carrier in p53 gene delivery. The carrier was shown to possess favorable plasmid binding and condensation ability with a hydrodynamic size and a zeta potential of 89.1 ± 1.8 nm and +9.1 ± 1.8 mV, respectively, at a mass ratio of 40. Using the human cervical carcinoma cell line HeLa as a model, carrier/p53 transfection has been demonstrated to improve the expression level of the p53 gene by qPCR and western blotting. After the successful p53 gene delivery, an obvious anti-proliferative effect was observed by the MTT method, Live/Dead staining and colony formation inhibition assays. The inhibition mechanism of cell proliferation was further shown to be associated with the induction of cell cycle arrest at the S phase and cell apoptosis through the activation of the mitochondria-dependent signaling pathway. Finally, the migration of tumor cells was restrained after p53 transfection, as elucidated by wound healing and Transwell migration assays. In summary, cholesterol-g-poly(amine-co-ester)-mediated p53 gene transfection could be a potential route to achieve p53-based gene therapy.


 Please wait while we load your content...
Please wait while we load your content...