Li2S–Embedded copper metal–organic framework cathode with superior electrochemical performance for Li–S batteries†
Abstract
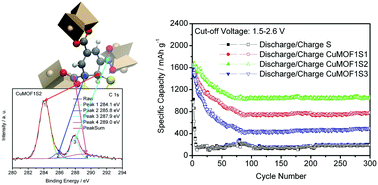
Li–S rechargeable batteries are desirable for electric transportation because of their low cost, environmental friendliness, and superior energy density. However, the Li–S system has yet to conquer the market owing to its drawbacks such as soluble polysulfide formation. To tackle this issue, we report herein a strategy based on the use of a micro-porous copper metal–organic framework named CuMOF as a host material for sulfur immobilization via chemical bonds between sulfur and the MOF structure. CuMOF has been obtained by simple chemical synthesis using copper salt and benzene-1,3,5-tricarboxylic acid as the ligand. Then, by the melt-diffusion method, sulfur-impregnated CuMOFS material is finally fabricated. As a cathode material for Li–S batteries, the CuMOFS cathode delivers a capacity of 1051.3 mA h g−1 after 300 cycles at a charge/discharge current density of 200 mA g−1. It is worth noting that the main cathode structure can retain more than 19.3 wt% sulfur due to the CuMOF host, which dramatically decreases capacity fading and improves coulombic efficiency during prolonged charge/discharge cycling. The XPS peaks of CuMOFS are assigned according to the information obtained from density functional theory (DFT) calculations. Linear scaling DFT structure relaxation for a model with >1000 atoms is carried out to exemplify the interactions between CuMOF framework and embedded Li2S. The excellent electrochemical performance can be due to firm confinement of sulfur/polysulfides because of strong chemical interactions between carbon and sulfur species in the CuMOF host.


 Please wait while we load your content...
Please wait while we load your content...