A novel fluorescent peptidyl probe for highly sensitive and selective ratiometric detection of Cd(ii) in aqueous and bio-samples via metal ion-mediated self-assembly†
Abstract
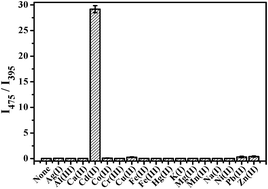
It is still a significant challenge to synthesize ratiometric fluorescent probes for Cd2+ ions in aqueous solutions and biosamples. In the present study, we synthesized a fluorescent peptidyl probe (1, pyrene-Cys-Gly-Pro-Cys-OH) based on a Cd2+-triggered self-assembling process for the ratiometric fluorescent detection of Cd2+ in aqueous solution, urine, and live cells. The peptidyl probe (1) consisting of a pyrene fluorophore and a tetrapeptide receptor did not self-assemble in aqueous solutions, resulting in significant pyrene monomer emission at 395 nm. Upon addition of Cd2+, the probes self-assembled into nanoparticles, showing significant enhancement of the excimer emission at 475 nm and a significant decrease in monomer emission at 395 nm, as revealed by fluorescence, dynamic light scattering, and transmission electron microscopy studies. Among 16 metal ions tested, only Cd2+ induced a sensitive ratiometric response. The ratiometric response showed an excellent linearity to Cd2+ ions, ranging from nanomolar to micromolar concentrations in aqueous solutions and urine samples. The detection limit for Cd2+ was 22 nM (R2 = 0.984) and the dissociation constant was measured to be 130 pM (R2 = 0.997) in aqueous solutions. Two thiol groups and a C-terminal carboxylic acid group of the peptide receptor of 1 play a critical role in binding with Cd2+. Furthermore, the probe penetrated live cells and detected intracellular Cd2+ by ratiometric responses. The specific metal ion-induced self-assembly of peptidyl probes could be a novel platform for the development of fluorescent detection methods for metal ions in aqueous samples and biosamples.


 Please wait while we load your content...
Please wait while we load your content...