Development of a selective and sensitive Ga3+ sensor for environmental safety: a comparative study of cyclohexyl and aromatic bis-sulphonamide fabricated glassy carbon electrodes†
Abstract
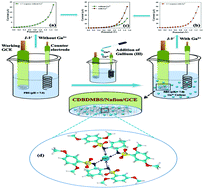
Our environment is becoming more polluted due to the commercial applications of toxic chemicals and heavy metal cations. So, for the sensing of heavy metal cations in aqueous solution N,N′-(cyclohexane-1,2-diyl)bis(2,5-dimethoxybenzenesulfonamide) CDBDMBS (3a) and N,N′-(1,2-phenylene)bis(2,5-dimethoxybenzenesulfonamide) PBDMBS (3b) were synthesized in this work. Spectroscopic techniques, such as UV/Vis spectroscopy, Fourier transform infrared (FTIR) spectroscopy, and nuclear magnetic resonance (1H-NMR & 13C-NMR) spectroscopy, and single crystal X-ray diffraction studies, were carried out for their structure elucidation. After that, a simple, cheap and reliable electrochemical technique was carried out in order to check the affinity of our newly designed chelating agents towards various heavy metal cations (Ag+, As3+, Au3+, Ce2+, Cr3+, Ga3+, Hg2+, Pb2+ and Y3+) using a Keithley electrometer. For this purpose CDBDMBS and PBDMBS were fabricated with conducting Nafion binder on a glassy carbon electrode (GCE) for the development of sensitive and selective electrochemical sensors to probe the heavy metal cations in a phosphate buffer phase (pH = 7.0) in a short response time, qualitatively and quantitatively. It was found that CDBDMBS was very selective with good affinity for gallium cations after a comparative study between CDBDMBS/Nafion/GCE and PBDMBS/Nafion/GCE. Gallium is one of the various heavy metal cations in our ecosystem as an environmental pollutant. The analytical performance of CDBDMBS/Nafion/GCE was found to be very interesting in terms of sensitivity, stability, linear dynamic range and improved electrochemical performance towards Ga3+ ions. The calibration plot is linear (r2 = 0.9006) over a large concentration range of Ga3+ ions (1.0 nM to 0.01 M) in addition to the sensitivity, limit of detection (at a SNR of 3) and limit of quantification that are found to be 3.955 × 10−3 μA μM−1 cm−2, 0.02 nM and 0.08 nM, respectively. This novel approach introduces a new and well organized system for the development of efficient sensors for toxic cationic electrochemicals in environmental and healthcare fields on a large scale. Therefore, this newly designed CDBDMBS/Nafion/GCE presents a cost effective compound that can be used as a feasible substitute for the detection and removal of Ga3+ions from water samples efficiently.


 Please wait while we load your content...
Please wait while we load your content...