Photochemical rearrangement reactions of bicyclic molecules that contain a cyclopropane ring†
Abstract
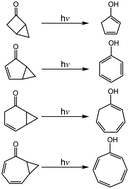
The mechanisms of photochemical rearrangement reactions are studied theoretically using a model system of bicyclic molecules that features a cyclopropane ring that is in proximity to a double bond moiety (such as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C or C
C or C ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group), using the M06-L method and the 6-311G(d,p) basis set. The theoretical findings show that intersystem crossing is vital to successfully interpret the mechanisms. This theoretical research also shows that the photoisomerization mechanism should proceed as follows: Rea-S0 → FC-T1 → Min-T1 → TS1-T1 → T1/S0 → Int1-S0 → TS2-S0 → Int2-S0 → TS3-S0 → Pro-S0. The theoretical evidence in this work confirms the experimental results for both [3.1.0] and [5.1.0] bicyclic molecules. The theoretical observations also predict that for both [2.1.0] and [4.1.0] systems, the quantum yields of the tautomeric keto forms are greater than those of the corresponding enol isomers.
O group), using the M06-L method and the 6-311G(d,p) basis set. The theoretical findings show that intersystem crossing is vital to successfully interpret the mechanisms. This theoretical research also shows that the photoisomerization mechanism should proceed as follows: Rea-S0 → FC-T1 → Min-T1 → TS1-T1 → T1/S0 → Int1-S0 → TS2-S0 → Int2-S0 → TS3-S0 → Pro-S0. The theoretical evidence in this work confirms the experimental results for both [3.1.0] and [5.1.0] bicyclic molecules. The theoretical observations also predict that for both [2.1.0] and [4.1.0] systems, the quantum yields of the tautomeric keto forms are greater than those of the corresponding enol isomers.


 Please wait while we load your content...
Please wait while we load your content...