Water-tolerant solid Lewis-acid sites: Baeyer–Villiger oxidation with hydrogen peroxide in the presence of gallium-based silica catalysts
Abstract
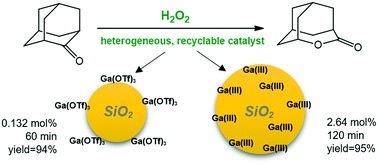
The Baeyer–Villiger oxidation of a model cyclic ketone, 2-adamantanone, using H2O2 as the oxidizing agent, was systematically studied in the presence of two types of heterogeneous catalysts based on gallium(III) triflate as a hydrolytically stable Lewis acid precursor. Active-phase silica with a bimodal pore-size distribution of mesopores was used as a solid carrier. The first type of catalyst was prepared by post-synthesis treatment with gallium(III) triflate using an impregnation procedure, while the second type assumed the use of gallium(III) triflate as a source of Ga catalytic centres incorporated into a silica structure in a one-pot synthesis. The structural parameters of both catalysts were determined. The extremely high activity of both catalysts in promoting the oxidation, allowing full conversion of the ketone with lactone formation in a very short time (20 min to 90 min, molar ratio ketone : 60 wt% aq. H2O2 = 1 : 2 at 70 °C), is reported for the first time. In the case of the use of the catalysts for oxidation in a one-pot synthesis, the amount of the catalyst was low (a ratio of 2.64 mol% of gallium to the ketone), but the second catalyst from post-synthesis treatment was used in truly catalytic amounts (0.132 mol%). The superactivity of this catalyst (very high TON 711) is related to the availability of all catalytic centres on the surface of the catalyst. Lewis and Brønsted acid sites in the described catalysts were found, and the participation of both in the conversion of the ketone was postulated. To show the application potential, the recycling studies of the catalysts were demonstrated.


 Please wait while we load your content...
Please wait while we load your content...