A highly selective fluorescence turn-on chemosensor for Zn2+, and its application in live cell imaging, and as a colorimetric sensor for Co2+: experimental and TD-DFT calculations†
Abstract
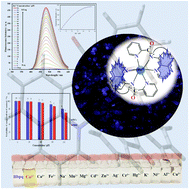
A small fluorogenic molecule Hbpq, N-(quinoline-8-yl)pyridine-2-carboxamide, for monitoring Zn(II) ions in vitro has been synthesized. Hbpq, prepared by a facile eco-friendly one-pot synthesis under mild reaction conditions with high efficiency, selectively exhibits a ∼120-fold increase in fluorescence intensity upon addition of half an equivalent of Zn2+. The chemoselective response of Hbpq to the Zn2+ ion in the presence of interfering ions in acetonitrile has also been examined. The results of the Job's plot experiment, the fluorescence and UV-vis titration, and ESI-MS, indicate that the binding stoichiometric ratio between Hbpq and Zn2+ in acetonitrile is 2 : 1. Single crystal X-ray structure determination of the zinc complex, synthesized independently, also confirms the formation of mononuclear Zn(bpq)2. In addition, Hbpq acts as a sensitive optical chemosensor for Co2+ ions in acetonitrile. The association constant (Ka) and limit of detection (LOD) for the Zn2+ and Co2+ complexes are also calculated. The cytotoxicity of Hbpq has been examined, and its applicability for visualizing intracellular Zn2+ has been demonstrated. Importantly, the MTT assay and cell imaging experiments show that Hbpq exhibits low cytotoxicity, good cell membrane permeability, and high potential for monitoring Zn2+ ions in vitro. The weak emission of Hbpq in MeCN solution is highly sensitive to the presence of water, being almost quenched, however this sensor behaves like a molecular light switch after entering the living cell and being surrounded by a hydrophobic environment. DFT/TD-DFT calculations have also been employed to support the binding mode and photophysical properties of Hbpq towards Zn2+ and the enhancement of its fluorescence by Zn2+ in living cells.


 Please wait while we load your content...
Please wait while we load your content...