Fixation of atmospheric nitrogen by nanodiamonds†
Abstract
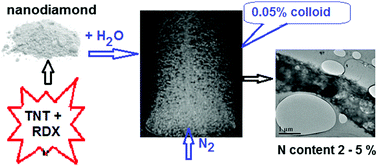
Very dilute aqueous colloids of a detonation-produced nanodiamond support the growth of fungi, which contain more nitrogen than that present in the nanodiamond as admixtures. We show that the extra nitrogen comes from the fixation of atmospheric N2, rather than from the concentration of nitrogen admixtures in the nanodiamond. N2 fixation can also be achieved using the colloids of an ordinary, nitrogen-free, synthetic diamond. The process (distinct from the known, extremely low-yield photolytic one) is made thermodynamically possible due to the lower (as proven by calorimetry) atomisation energy of carbon in nanoparticles. It converts nanodiamonds into O-, N- and H-enriched solids of variable composition, which lose mass exothermically upon heating in an argon atmosphere, yielding CO2, H2O, NO and NO2 through internal oxidation.


 Please wait while we load your content...
Please wait while we load your content...