One-pot synthesis of carbon-coated Fe3O4 nanoparticles with tunable size for production of gasoline fuels†
Abstract
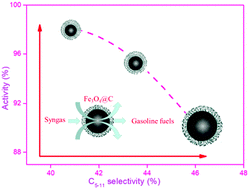
Carbon-coated Fe3O4 nanoparticles with tunable size were fabricated through a facile one-pot solvothermal method and employed as catalysts for Fischer–Tropsch (FT) synthesis to optimize the product distribution of hydrocarbons. Each spherical nanoparticle (Fe3O4@C) was observed to consist of a core–shell structure with an Fe3O4 core and a carbon shell. Furthermore, the Fe3O4 particle size can be effectively tailored by altering the initial dosage of glucose during catalyst preparation. Fe3O4@C with a suitable structure exhibited notable catalytic activity (CO conv. over 98%), high C5–C11 selectivity (46.6%) and superior stability in the FT synthesis. It was proposed that Fe3O4@C with a small particle size facilitated the formation of more active sites, which are responsible for the enhanced catalytic activity. However, the increase in particle and pore size promoted the selectivity towards C5+ hydrocarbons and lower olefins. This may provide a novel route to directly synthesize gasoline fuels during FTS.


 Please wait while we load your content...
Please wait while we load your content...