Development of a sensor for trivalent iron: AHP fixed on mesoporous silica†
Abstract
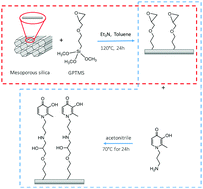
Since the emergence of deferiprone as an iron(III) chelating drug, hydroxypyridinones have been intensively explored due to their high affinity for trivalent metal ions and ability to form complexes at physiological pH with low toxicity. For instance, they have been employed as carriers of La(III) in therapy for bone diseases and to remove Gd(III) from Gd-based contrast agents. We believe that a pyridinone-based sensor can be useful for iron(III) monitoring. Herein, we present a novel chelating solid-phase, resulting from the functionalization of a mesoporous silica MCM-41, with the hydroxypyridinone N(3′-aminopropyl)-3-hydroxy-2-methyl-4-pyridinone (AHP) as the active site. The physico-chemical characterization of the new solid-state device, named AHP-MCM41@, demonstrates that the AHP moiety is covalently anchored on the silica surface; the active site concentration was found to be around 0.4 mmol g−1. Furthermore, its sorption of Fe(III) from aqueous solution is rather rapid. The soluble AHP forms a rich variety of complexes with iron(III), which is typical of all analogous O,O donor ligands, and is dominated by M : L = 1 : 3 complexes. Could complexes with such high stoichiometry can be retained in the solid phase? The answer is yes. The existence of these different species in the solid phase is evidenced by the sorption isotherm results and from sorption experiments as a function of solution pH and also in the presence of a competitive ligand. Furthermore, the existence of complexes in the solid phase was demonstrated via solid vis-spectrophotometry, where these species showed identical colour variations to those formed in solution. Such complexes in the solid phase have never been reported in the literature. Additionally, the intense colour of the solid phase in the presence of iron(III) is promising for naked eye detection.
- This article is part of the themed collection: Equilibrium Solution Coordination Chemistry


 Please wait while we load your content...
Please wait while we load your content...