Ferrocene amphiphilic D–π–A dyes: synthesis, redox behavior and determination of band gaps†
Abstract
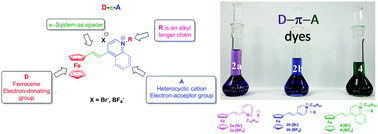
We report the synthesis of a series of ferrocene amphiphilic donor–π–acceptor dyes, with the general formula (Fc-CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-HetNC16H33)+ X− [where: Fc behaves as the donor group, a double bond as the π bridge, and 2-,4-pyridinium and 4-quinolinium as the potent acceptor groups (2a–b and 4, X = Br− or BF4−)], in good overall yields. Together with their neutral counterparts (6a–b and 7), the photophysical and electrochemical properties of these compounds were investigated by means of UV-Vis spectroscopy and cyclic voltammetry. The optical and electrochemical band gaps of these dyes were calculated, which indicated that 4 has the lowest bandgap value. Time-dependent DFT calculations indicate that the lowest energy absorption band displayed for these compounds has mainly metal-to-ligand charge transfer character, with the HOMO–LUMO electronic transition being the main contribution.
CH-HetNC16H33)+ X− [where: Fc behaves as the donor group, a double bond as the π bridge, and 2-,4-pyridinium and 4-quinolinium as the potent acceptor groups (2a–b and 4, X = Br− or BF4−)], in good overall yields. Together with their neutral counterparts (6a–b and 7), the photophysical and electrochemical properties of these compounds were investigated by means of UV-Vis spectroscopy and cyclic voltammetry. The optical and electrochemical band gaps of these dyes were calculated, which indicated that 4 has the lowest bandgap value. Time-dependent DFT calculations indicate that the lowest energy absorption band displayed for these compounds has mainly metal-to-ligand charge transfer character, with the HOMO–LUMO electronic transition being the main contribution.
- This article is part of the themed collection: Celebrating recent chemical science in Mexico


 Please wait while we load your content...
Please wait while we load your content...