A Michael addition–cyclization-based switch-on fluorescent chemodosimeter for cysteine and its application in live cell imaging†
Abstract
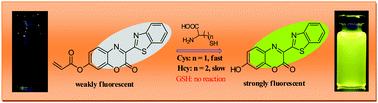
Based on a conjugate addition/intramolecular cyclization sequence, we designed and synthesized a fast response fluorescent probe, BTAC (benzothiazol-azacoumarin), for the discriminative detection of cysteine (Cys). The reaction of cysteine with BTAC results in the cleavage of the acrylate moiety from BTAC, thereby producing BTAC-OH, with a remarkable fluorescence enhancement at 560 nm. The probe exhibits high sensitivity and selectivity toward cysteine over homocysteine and glutathione and the detection limit reached as low as 124 nM for cysteine. The addition of Cys resulted in the color of the solution of BTAC changing from colorless to greenish yellow under the simulation of physiological conditions and BTAC could serve as a “naked-eye” indicator. The structure of BTAC was established by computational DFT (density functional theory) calculation and time dependent density functional theory (TDDFT) calculations were performed to demonstrate the electronic properties of BTAC and its product, BTAC-O−. Finally, the probe was successfully applied for the fluorescence bioimaging of cysteine owing to its photostability and low cytotoxicity.


 Please wait while we load your content...
Please wait while we load your content...