Ruthenium carbonyl complexes with pyridylalkanol ligands: synthesis, characterization and catalytic properties for aerobic oxidation of secondary alcohols†
Abstract
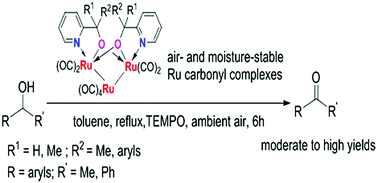
Reaction of Ru3(CO)12 with pyridylalkanol ligands PyC(CH2)4OH (L1H), PyC(CH2)5OH (L2H) and PyCR1R2OH (R1 = R2 = CH3 (L3H); R1 = CH3, R2 = C6H5 (L4H); R1 = H, R2 = C6H5 (L5H); R1 = H, R2 = 4-CH3C6H4 (L6H); R1 = H, R2 = 4-OMeC6H4 (L7H); R1 = H, R2 = 4-ClC6H4 (L8H); R1 = H, R2 = 4-BrC6H4 (L9H); R1 = H, R2 = 4-CF3C6H4 (L10H)) in refluxing xylene afforded the bis-chelate ruthenium carbonyl complexes [(Ln)2Ru3(CO)8] (n = 1 (1a); n = 2 (1b); n = 3 (1c); n = 4 (1d); n = 5 (1e); n = 6 (1f); n = 7 (1g); n = 8 (1h); n = 9 (1i); n = 10 (1j)), respectively. All the novel ruthenium complexes were fully characterized by NMR, elemental analyses and IR spectra and the molecular structures of 1a, 1c, 1e, 1g and 1i were further determined by single crystal X-ray diffraction analysis. In the presence of TEMPO (TEMPO = 2,2,6,6-tetramethyl-1-piperidinyloxyl), these trirhenium carbonyl clusters displayed high reactivity for aerobic oxidation of secondary alcohols to give the corresponding ketonic compounds in good to excellent yield using ambient air as the source of oxidant.


 Please wait while we load your content...
Please wait while we load your content...