Facile microwave synthesis, structural diversity and herbicidal activity of six novel alkaline-earth metal complexes (AECs) based on skeletal isomerization chlorophenoxyacetic acids†
Abstract
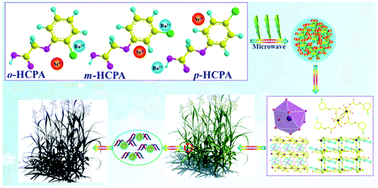
Six novel AECs of [Sr(o-CPA)2(H2O)4]·(o-CPA) 1, [Ba(o-CPA)2(H2O)]·H2O 2, [Sr(m-CPA)2(H2O)4]·(m-CPA) 3, [Ba(m-CPA)2(H2O)4]2·2(m-CPA)·H2O 4, [Sr(p-CPA)2(H2O)] 5 and [Ba(p-CPA)2(H2O)] 6 (o-CPA = 2-chlorophenoxyacetic acid, m-CPA = 3-chlorophenoxyacetic acid and p-CPA = 4-chlorophenoxyacetic acid) were synthesized by a facile microwave-assistant reaction. The solid-state structures were well established by X-ray crystallography and routine analyses of Fourier transform infrared, elemental analysis, field emission scanning electron microscope (FESEM) and thermogravimetric analysis. The structure data reveal that complexes of 1, 3 and 4 are one-dimensional chains with a zigzag arrangement, while 2, 5 and 6 are (4, 4) rhomboid two-dimensional grid structures. It is noticed that the carboxyl group in these complexes displays multiple coordination modes of μ2-η1:η1 (1, 2, 3, 4, 5 and 6), μ3-η1:η2 (1, 2, 3 and 4), μ2-η1:η2 (2), μ3-η2:η2 (5 and 6). Interestingly, the oxygen atoms of flexible –OCH2– in phenoxy groups take part in coordination behavior with metal centers through the rotation of the C–O and C–C bonds in complexes 2, 5 and 6, while only carboxyl group coordination can be found in complexes 1, 3 and 4. FESEM images indicate that the surface appearances of complexes are totally different from ligands after coordination. All ligands and complexes were evaluated for activity as plant-growth inhibitors against Amaranth (Amaranthus spp.) and barnyard grass. Compared with ligands, complexes 1, 3 and 4 exhibited better response index (RI) values of Shoot elongation against barnyard grass. Moreover, complexes 1, 3 and 4 demonstrated a higher inhibitory activity than 2, 5 and 6. It is significant to develop a new kind of environmentally-friendly herbicide based on these kinds of complexes for their low toxicity and high efficiency.


 Please wait while we load your content...
Please wait while we load your content...