Synthesis and coordination studies of 5-(4′-carboxyphenyl)-10,15,20-tris(pentafluorophenyl)porphyrin and its pyrrolidine-fused chlorin derivative†
Abstract
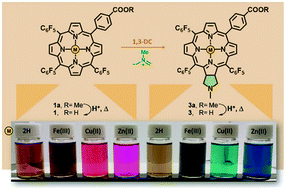
The introduction of a carboxylate function into porphyrins allows a variety of modifications, including coordination and conjugation, which are central to enhance the efficiency of macrocycles in photonic materials and biological applications. Herein, a synthetic strategy to obtain 5-(4′-carboxyphenyl)-10,15,20-tris(pentafluorophenyl)porphyrin and its pyrrolidine-fused chlorin derivative was developed by the 1,3-dipolar cycloaddition of a carbomethoxyphenyl substituted porphyrin with an azomethine ylide, followed by hydrolysis under thermal acidic conditions. The 1,3-dipolar cycloaddition of the carbomethoxyphenyl porphyrin with N-methyl nitrone was also performed to give an isomeric mixture of isoxazolidine-fused chlorins, revealing lower selectivity and lower yields; in addition, retrocycloaddition of the isoxazolidine-fused chlorins was observed under the hydrolysis conditions. The resulting carboxyphenyl macrocycles were characterized using 1H and 19F NMR, ESI-MS and SC-XRD for 5-(4′-carboxyphenyl)-10,15,20-tris(pentafluorophenyl)porphyrin. In order to study the influence of coordination to a metal ion on the electronic properties of carboxyphenyl substituted porphyrins, a series of metal complexes of 5-(4′-carboxyphenyl)-10,15,20-tris(pentafluorophenyl)porphyrin and its pyrrolidine-fused chlorin derivative were synthesized by microwave-mediated metallation with Fe(III), Cu(II) and Zn(II) salts. EPR spectroscopy was particularly relevant to the characterization of the Cu(II) complexes of both macrocycles and to study the coordination chemistry of these ligands with Cu(II) ions.
- This article is part of the themed collection: Equilibrium Solution Coordination Chemistry


 Please wait while we load your content...
Please wait while we load your content...