Rhodium(triphenylphosphine)carbonyl-2,4-dioxo-3-pentyl-4-decanyloxybenzoate: synthesis, electrochemistry and oxidative addition kinetics†
Abstract
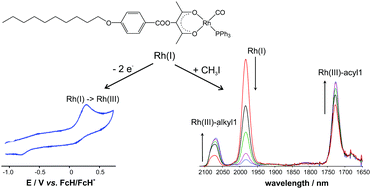
The synthesis, electrochemistry and oxidative addition kinetics are presented for a new [Rh(β-diketonato)(CO)(PPh3)] complex (2), of rhodium metal complexed with a β-diketonato ligand, β-L1 = (CH3COC(C10H21OC6H4COO)COCH3)−, containing a long and sterically large chain (Rα = C10H21OC6H4COO) substituted at the α position. This rhodium triphenylphosphine complex (2), [Rh(β-L1)(CO)(PPh3)], was subsequently converted from a rhodium(I) to a rhodium(III) complex, by chemical and electrochemical oxidation. The kinetics of the chemical conversion from RhI to RhIII, which was obtained by the oxidative addition reaction [Rh(β-L1)(CO)(PPh3)] + CH3I, demonstrated that the reaction occurs in two reaction steps, with a RhIII-alkyl species as the main reaction product. The sterically large and long chain (Rα = C10H21OC6H4COO) at the α-position of the β-diketonato ligand, did not at all affect either the second order reaction rate constant (k1) of the first oxidative addition step of the [Rh(β-L1)(CO)PPh3] + CH3I reaction, nor the value of the oxidation potential (Epa) of the electrochemical oxidation of [RhI(β-L1)(CO)PPh3] to a RhIII-species. This was proven via a literature study, by comparing the k1 and Epa values from this study with the respective rate constants (k1) and oxidation potentials (Epa) of a wide variety of related [Rh(β-diketonato)(CO)(PPh3)] complexes without such a Rα substituent at the α position.


 Please wait while we load your content...
Please wait while we load your content...