Toxic heavy metal – Pb, Cd, Sn – complexation by the octadentate hydroxypyridinonate ligand archetype 3,4,3-LI(1,2-HOPO)†
Abstract
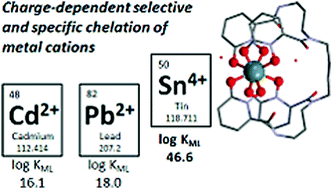
The toxicity of heavy metals such as lead (Pb), cadmium (Cd) and tin (Sn) has long been known but accidental exposures of large populations to these elements remain unfortunately a topical issue. Chelating agents against Pb, Cd, or Sn poisoning are still limited to classical ligands such as ethylenediamine tetraacetic acid (EDTA). Here, we evaluate the ability of 3,4,3-LI(1,2-HOPO) (L4−), an octadentate ligand currently under evaluation for actinide decorporation, to bind Pb, Cd and Sn metal ions in aqueous solutions. This ligand forms 1 : 1 complexes with Pb(II), Cd(II) and Sn(IV) as well as bimetallic 2 : 1 species with Pb(II) and Cd(II), all of which were characterized by high resolution mass spectrometry and spectrophotometric titrations. The 3,4,3-LI(1,2-HOPO) ligand exhibits an extreme affinity for Sn(IV) ions (log β110 > 40), with the complex remaining stable from highly acidic conditions to alkaline media (from 3 M HCl to pH 8). Single crystals of the neutral complex [SnIV3,4,3-LI(1,2-HOPO)·3H2O] were obtained and its structure determined, revealing a chiral conformation. Although not initially designed for hexacoordinated metals such as Pb(II) and Cd(II), the octadentate ligand shows promise for the in vitro or in vivo sequestration of toxic heavy metals, as evidenced by decorporation experiments performed in mice contaminated with 210Pb(II) and treated with either EDTA or 3,4,3-LI(1,2-HOPO).
- This article is part of the themed collection: Equilibrium Solution Coordination Chemistry


 Please wait while we load your content...
Please wait while we load your content...