Synthesis and structural, photophysical, electrochemical redox and axial ligation properties of highly electron deficient perchlorometalloporphyrins and selective CN− sensing by Co(ii) complexes†
Abstract
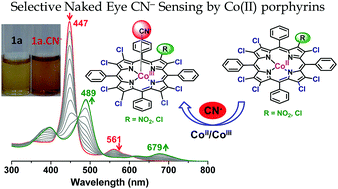
A straightforward synthetic route has been adopted to synthesize highly nonplanar electron deficient perchlorometallo-porphyrins. Herein, we report the synthesis and characterization of MTPP(NO2)Cl7 where M = CoII, NiII, CuII and ZnII. Further, we examined their optical and electrochemical redox properties and the results are compared with MTPPCl8. MTPP(NO2)Cl7 exhibited red-shifted (∼10–15 nm) absorption spectra relative to MTPPCl8 due to the strong electron withdrawing nature of the nitro group. Mixed β-substitution alters the electrochemical redox properties to such an extent that an appreciable increase in the anodic shift in reduction potential (200–300 mV) is observed for MTPP(NO2)Cl7 relative to MTPPCl8 whereas only a minimal shift (15–50 mV) in the oxidation potential is observed. Nonplanarity of the macrocyclic core was investigated by single crystal X-ray analysis and DFT calculations. A higher ΔCβ (0.706 Å) for 1d as compared to 2d (0.642 Å) undoubtedly signifies nonplanarity induced by the nitro group. To substantiate the effect of mixed substitution, we performed axial ligation studies of Zn(II) complexes with nitrogenous bases and basic anions and found higher log β2 values as well as a linear relation between log β2 and pKa as compared to perbromoporphyrins. Highly electron deficient β-substituted Co(II) porphyrins (1a and 2a) were utilized as novel sensors for selective rapid visual detection of CN− ions.
- This article is part of the themed collection: Equilibrium Solution Coordination Chemistry


 Please wait while we load your content...
Please wait while we load your content...