Metallic CuNPs confined in hollow silicalite-1: excellent catalytic efficiency in p-nitrophenol reduction†
Abstract
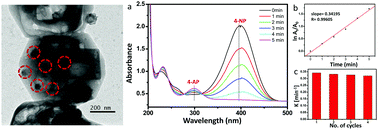
Copper nanoparticles (CuNPs) confined in hollow silicalite-1 powders were synthesized by a selective “desilication–recrystallization” method using tetraethlyorthosilicate (TEOS), tetrapropylammonium hydroxide (TPAOH) and cupric chloride (CuCl2) as precursors. The synthesized product was characterized by XRD, Raman, XPS, FESEM, TEM and N2 adsorption–desorption studies. The presence of CuNPs of size 10–40 nm in the hollow silicalite-1 was confirmed by TEM. The BET surface area of the powders was 247 m2 g−1 composed of micropores and mesopores. The prepared CuNPs confined in hollow silicalite-1 showed excellent catalytic performance for the reduction of 4-nitrophenol (4-NP) to 4-aminophenol (4-AP) with apparent rate constant and activity parameter values of 5.6 × 10−3 s−1 and 44.09 s−1 g−1, respectively for 1 mg of catalyst.


 Please wait while we load your content...
Please wait while we load your content...