Simple tuning of the luminescence properties of the double rollover cycloplatinated(ii) structure by halide ligands†
Abstract
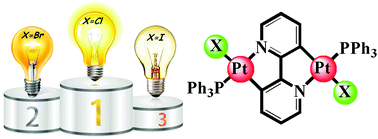
The present study highlights the effects of anionic ligands, i.e., halide or trifluoroacetate, on the emission properties of cycloplatinated(II) complexes with a double rollover structure. In fact, it has been proven that the halide ligands can tune the brightness of organometallic compounds as “organic light emitting diodes (OLEDs) emitters”. Thus, a series of closely related platinum (II) complexes with general formula of [Pt2(μ-bpy-2H)(X)2(PPh3)2] has been prepared by incorporating a double rollover cyclometalated ligand (μ-bpy-2H), two triphenylphosphine ligands and either two trifluoroacetate ligands (X = CF3COO, 3) or two halide ligands (X = Cl, 4a, Br, 4b, or I, 4c). The complexes were characterized by multi-nuclear 1H and 31P NMR spectroscopy and the structure of the complex [Pt2(μ-bpy-2H)(CF3COO)2(PPh3)2] (3) was typically determined by single crystal X-ray crystallography. The luminescence properties of the newly synthesized complexes were investigated at 298 K and 77 K over the wide range of 295–505 nm for excitation wavelengths. Although the emission bands appeared at almost the same wavelengths for the luminescent complexes, differences in both quantum yield and brightness were observed depending on the nature of X. In fact, the intrinsic ligand field of the X ligands has a substantial impact on the platinum-ligand bond lengths. Complex 4c (having iodo ligands with the lowest ligand field) exhibits the highest thermal population for the higher lying metal-centered electronic state (d–d* character) and consequently the highest non-radiative deactivation rate, which remarkably quenches the emission.


 Please wait while we load your content...
Please wait while we load your content...