Near infra-red dyes based on pyrene aza-BODIPYs†
Abstract
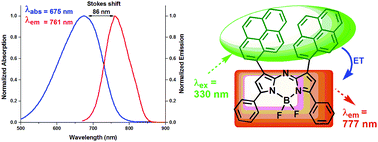
Pyrene substituted donor–acceptor type NIR aza-BODIPYs were designed and synthesized. The 1,7-positions of the aza-BODIPY core were substituted by pyrene moieties and the 3,5-positions were substituted with phenyl/2-thienyl or anisyl groups in the aza-BODIPYs. The effect of substituents on their photophysical and electrochemical properties was studied; also, computational studies were carried out for the target aza-BODIPYs. These near infrared dyes displayed the lowest energy absorption maxima in the range of 670–738 nm; whereas, their emission maxima were centered on ≈764 nm. The absorption and emission spectra of the aza-BODIPYs were significantly red shifted (72–94 nm) as compared to the parent tetra-phenylaza-BODIPY. Fluorescence studies suggested effective energy transfer (up to 65%) from donor groups to the aza-BODIPY core in all the compounds. TD-DFT studies indicated significant electronic interactions between energy donor groups and the aza-BODIPY core in all the aza-BODIPYs. The values of HOMO–LUMO gap (ΔE) calculated from cyclic voltammetry data were comparable with those obtained from DFT studies.


 Please wait while we load your content...
Please wait while we load your content...