Is hydroxypyridonate 3,4,3-LI(1,2-HOPO) a good competitor of fetuin for uranyl metabolism?†
Abstract
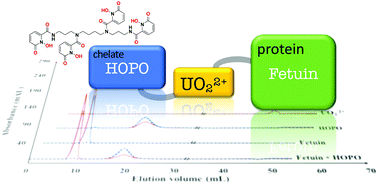
Uranium is widespread in the environment, resulting both from natural occurrences and anthropogenic activities. Its toxicity is mainly chemical rather than radiological. In the blood it is transported as uranyl UO22+ cation and forms complexes with small ligands like carbonates and with some proteins. From there it reaches the skeleton, its main target organ for accumulation. Fetuin is a serum protein involved in biomineralization processes, and it was demonstrated to be the main UO22+-binder in vitro. Fetuin's life cycle ends in bone. It is thus suspected to be a key protagonist of U accumulation in this organ. Up to now, there has been no effective treatment for the removal of U from the body and studies devoted to the interactions involving chelating agents with both UO22+ and its protein targets are lacking. The present work aims at studying the potential role of 3,4,3-LI(1,2-HOPO) as a promising chelating agent in competition with fetuin. The apparent affinity constant of 3,4,3-LI(1,2-HOPO) was first determined, giving evidence for its very high affinity similar to that of fetuin. Chromatography experiments, aimed at identifying the complexes formed and quantifying their UO22+ content, and spectroscopic structural investigations (XAS) were carried out, demonstrating that 3,4,3-LI(1,2-HOPO) inhibits/limits the formation of fetuin–uranyl complexes under stoichiometric conditions. But surprisingly, possible ternary complexes stable enough to remain present after the chromatographic process were identified under sub-stoichiometric conditions of HOPO versus fetuin. These results contribute to the understanding of the mechanisms accounting for U residual accumulation despite chelation therapy after internal contamination.


 Please wait while we load your content...
Please wait while we load your content...