Nickel(ii) inhibits the oxidation of DNA 5-methylcytosine in mammalian somatic cells and embryonic stem cells
Abstract
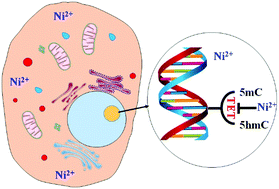
Nickel is found widely in the environment. It is an essential microelement but also toxic. However, nickel displays only weak genotoxicity and mutagenicity. Exploration of the epigenetic toxicity of nickel is extremely interesting. Iron(II)- and 2-oxoglutarate-dependent Tet dioxygenases are a class of epigenetic enzymes that catalyze the oxidation of DNA 5-methylcytosine (5mC). Thus, they are critical for DNA demethylation and, importantly, are involved with nuclear reprogramming, embryonic development, and regulation of gene expression. Here, we demonstrated that nickel(II) dramatically inhibits Tet proteins-mediated oxidation of DNA 5mC in cells ranging from somatic cell lines to embryonic stem cells, as manifested by the consistent observation of a significant decrease in 5-hydroxymethylcytosine, a critical intermediate resulting from the oxidation of 5mC. The inhibitory effects of nickel(II) were concentration- and time-dependent. Using HEK293T cells overexpressing Tet proteins and ascorbic acid-stimulated Tet-proficient ES cells, we observed that nickel(II) significantly reduced DNA demethylation at the global level. Interestingly, we also showed that nickel(II) might affect the naïve or ground state of pluripotent embryonic stem cells. Here we show, for the first time, that nickel(II) represses the oxidation of DNA 5mC and potentially alters the Tet proteins-regulated DNA methylation landscape in human cells. These findings provide new insights into the epigenetic toxicology of nickel.
- This article is part of the themed collection: Metallomics Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...