Iron–sulfur cluster biosynthesis and trafficking – impact on human disease conditions
Abstract
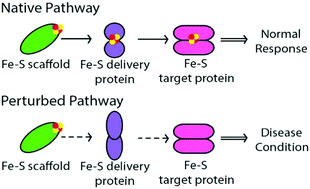
Iron–sulfur clusters (Fe–S) are one of the most ancient, ubiquitous and versatile classes of metal cofactors found in nature. Proteins that contain Fe–S clusters constitute one of the largest families of proteins, with varied functions that include electron transport, regulation of gene expression, substrate binding and activation, radical generation, and, more recently discovered, DNA repair. Research during the past two decades has shown that mitochondria are central to the biogenesis of Fe–S clusters in eukaryotic cells via a conserved cluster assembly machinery (ISC assembly machinery) that also controls the synthesis of Fe–S clusters of cytosolic and nuclear proteins. Several key steps for synthesis and trafficking have been determined for mitochondrial Fe–S clusters, as well as the cytosol (CIA – cytosolic iron–sulfur protein assembly), but detailed mechanisms of cluster biosynthesis, transport, and exchange are not well established. Genetic mutations and the instability of certain steps in the biosynthesis and maturation of mitochondrial, cytosolic and nuclear Fe–S cluster proteins affects overall cellular iron homeostasis and can lead to severe metabolic, systemic, neurological and hematological diseases, often resulting in fatality. In this review we briefly summarize the current molecular understanding of both mitochondrial ISC and CIA assembly machineries, and present a comprehensive overview of various associated inborn human disease states.
- This article is part of the themed collections: Recent Review Articles and Metallomics 2018 Most Downloaded Articles


 Please wait while we load your content...
Please wait while we load your content...