An evolutionary transcriptomics approach links CD36 to membrane remodeling in replicative senescence†
Abstract
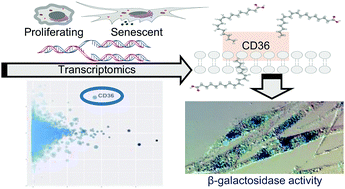
Cellular senescence, the irreversible ceasing of cell division, has been associated with organismal aging, prevention of cancerogenesis, and developmental processes. As such, the evolutionary basis and biological features of cellular senescence remain a fascinating area of research. In this study, we conducted comparative RNAseq experiments to detect genes associated with replicative senescence in two different human fibroblast cell lines and at different time points. We identified 841 and 900 genes (core senescence-associated genes) that are significantly up- and downregulated in senescent cells, respectively, in both cell lines. Our functional enrichment analysis showed that downregulated core genes are primarily involved in cell cycle processes while upregulated core gene enrichment indicated various lipid-related processes. We further demonstrated that downregulated genes are significantly more conserved than upregulated genes. Using both transcriptomics and genetic variation data, we identified one of the upregulated, lipid metabolism genes, CD36, as an outlier. We found that overexpression of CD36 induces a senescence-like phenotype and, further, the media of CD36-overexpressing cells alone can induce a senescence-like phenotype in proliferating young cells. Moreover, we used a targeted lipidomics approach and showed that phosphatidylcholines accumulate during replicative senescence in these cells, suggesting that upregulation of CD36 could contribute to membrane remodeling during senescence. Overall, these results contribute to the understanding of evolution and biology of cellular senescence and identify several targets and questions for future studies.


 Please wait while we load your content...
Please wait while we load your content...