Cooperative effects of inorganic and organic structure-directing agents in ZSM-5 crystallization†
Abstract
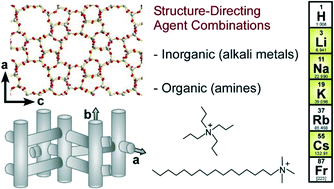
Zeolite crystallization occurs in the presence of inorganic and/or organic structure-directing agents (SDAs) that facilitate the formation of microporous crystals with various pore geometries. One of the most common zeolites is ZSM-5 (MFI type), which is used as a catalyst and sorbent in a wide range of industrial applications. One of the challenges with ZSM-5 synthesis is to identify new and inexpensive organic SDAs that can tailor the physicochemical properties of the final product. The most frequently used SDA in ZSM-5 synthesis is tetrapropylammonium (TPA); however, recent studies have shown that the surfactant cetyltrimethylammonium (CTA) can be used as an alternative SDA. Here, we examine the effects of dual structure-directing agents using CTA and TPA as organic SDAs in combination with a variety of alkali metals as inorganic SDAs. Our findings reveal that the selection of SDA combinations has a significant impact on the kinetics of ZSM-5 crystallization, as well as the properties of the resulting crystals. Notably, we show that TPA/Na and CTA/K are optimal combinations of SDAs that can markedly alter the size, morphology, and aluminum distribution in ZSM-5. Using a combination of experiments and molecular modeling, we explore the use of CTA as an alternative organic SDA for zeolite MFI and show that we can achieve smaller crystals (ca. 600 nm) in similar time (<24 h) as syntheses employing TPA. Given the relatively low cost of CTA and its high market availability compared to other organic SDAs, these findings have the potential to impact commercial production of ZSM-5 for a variety of applications.
- This article is part of the themed collections: 2018 MSDE Hot Articles, MSDE Emerging Investigators 2018 and Advances in Directed Self-Assembly


 Please wait while we load your content...
Please wait while we load your content...