Automated microfluidic droplet sampling with integrated, mix-and-read immunoassays to resolve endocrine tissue secretion dynamics†
Abstract
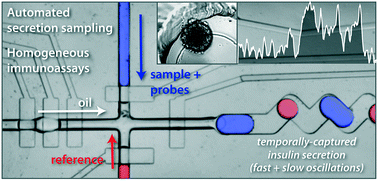
A fully automated droplet generation and analysis device based on pressure driven push-up valves for precise pumping of fluid and volumetric metering has been developed for high resolution hormone secretion sampling and measurement. The device consists of a 3D-printer templated reservoir for single cells or single tissue culturing, a Y-shaped channel for reagents and sample mixing, a T-junction channel for droplet formation, a reference channel to overcome drifts in fluorescence signal, and a long droplet storage channel allowing incubation for homogeneous immunoassays. The droplets were made by alternating peristaltic pumping of aqueous and oil phases. Device operation was automated, giving precise control over several droplet parameters such as size, oil spacing, and ratio of sample and reference droplets. By integrating an antibody–oligonucleotide based homogeneous immunoassay on-chip, high resolution temporal sampling into droplets was combined with separation-free quantification of insulin secretion from single islets of Langerhans using direct optical readout from the droplets. Quantitative assays of glucose-stimulated insulin secretion were demonstrated at 15 second temporal resolution while detecting as low as 10 amol per droplet, revealing fast insulin oscillations that mirror well-known intracellular calcium signals. This droplet sampling and direct optical analysis approach effectively digitizes the secretory time record from cells into droplets, and the system should be generalizable to a variety of cells and tissue types.
- This article is part of the themed collection: Lab on a Chip Recent HOT Articles


 Please wait while we load your content...
Please wait while we load your content...