Microfluidic filter device with nylon mesh membranes efficiently dissociates cell aggregates and digested tissue into single cells†
Abstract
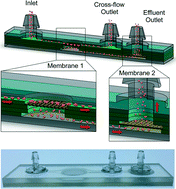
Tissues are increasingly being analyzed at the single cell level in order to characterize cellular diversity and identify rare cell types. Single cell analysis efforts are greatly limited, however, by the need to first break down tissues into single cell suspensions. Current dissociation methods are inefficient, leaving a significant portion of the tissue as aggregates that are filtered away or left to confound results. Here, we present a simple and inexpensive microfluidic device that simultaneously filters large tissue fragments and dissociates smaller aggregates into single cells, thereby improving single cell yield and purity. The device incorporates two nylon mesh membranes with well-defined, micron-sized pores that operate on aggregates of different size scales. We also designed the device so that the first filtration could be performed under tangential flow to minimize clogging. Using cancer cell lines, we demonstrated that aggregates were effectively dissociated using high flow rates and pore sizes that were smaller than a single cell. However, pore sizes that were less than half the cell size caused significant damage. We then improved results by passing the sample through two filter devices in series, with single cell yield and purity predominantly determined by the pore size of the second membrane. Next, we optimized performance using minced and digested murine kidney tissue samples, and determined that the combination of 50 and 15 μm membranes was optimal. Finally, we integrated these two membranes into a single filter device and performed validation experiments using minced and digested murine kidney, liver, and mammary tumor tissue samples. The dual membrane microfluidic filter device increased single cell numbers by at least 3-fold for each tissue type, and in some cases by more than 10-fold. These results were obtained in minutes without affecting cell viability, and additional filtering would not be required prior to downstream applications. In future work, we will create complete tissue analysis platforms by integrating the dual membrane microfluidic filter device with additional upstream tissue processing technologies, as well as downstream operations such as cell sorting and detection.
- This article is part of the themed collection: Lab on a Chip Recent HOT Articles


 Please wait while we load your content...
Please wait while we load your content...