Abstract
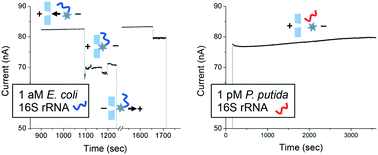
A nucleic acid amplification-free, optics-free platform has been demonstrated for sequence-specific detection of Escherichia coli (E. coli) 16S rRNA at 1 aM (10−18 M) against a 106-fold (1 pM) background of Pseudomonas putida (P. putida) RNA. This work was driven by the need for simple, rapid, and low cost means for species-specific bacterial detection at low concentration. Our simple, conductometric sensing device functioned by detecting blockage of a nanopore fabricated in a sub-micron-thick glass membrane. Upon sequence-specific binding of target 16S rRNA, otherwise charge-neutral, PNA oligonucleotide probe-polystyrene bead conjugates become electrophoretically mobile and are driven to the glass nanopore of lesser diameter, which is blocked, thereby generating a large, sustained and readily observable step decrease in ionic current. No false positive signals were observed with P. putida RNA when this device was configured to detect E. coli 16S rRNA. Also, when a universal PNA probe complementary to the 16S rRNA of both E. coli and P. putida was conjugated to beads, a positive response to rRNA of both bacterial species was observed. Finally, the device readily detected E. coli at 10 CFU mL−1 in a 1 mL sample, also against a million-fold background of viable P. putida. These results suggest that this new device may serve as the basis for small, portable, low power, and low-cost systems for rapid detection of specific bacterial species in clinical samples, food, and water.


 Please wait while we load your content...
Please wait while we load your content...