UV photochemical vapor generation of noble metals (Au, Ir, Pd, Pt and Rh): a feasibility study using inductively coupled plasma mass spectrometry and seawater as a test matrix
Abstract
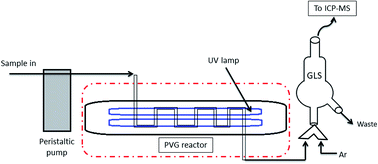
The results originated from the application of a simple and straightforward photochemical reactor to generate volatile species from noble metals (Au, Ir, Pd, Pt and Rh) are presented. Seawater samples were spiked with known amounts of the analytes and mixed with formic acid, followed by irradiation of the solution at 253.7 nm. The irradiated solution was driven to a gas–liquid separator and the volatile species were carried by an argon stream for detection using inductively coupled plasma mass spectrometry. Parameters such as formic acid concentration, carrier gas flow rate, UV irradiation period and RF power were evaluated. The optimum conditions were determined as 20% (v/v) formic acid, 1.0 L min−1 Ar, 45 s of UV exposure for Au, Pd, Pt and Rh or 120 s for Ir and 1300 W ICP RF power. Evaluation of potential interferences has shown that the addition of Cu2+ at ppm levels improved the overall efficiency of the photochemical process for Ir and Rh. Hence, Cu2+ was added as a modifier prior to the quantitation of Ir and Rh. Three seawater samples collected locally were used to evaluate the ability to carry out quantitative analysis of the investigated elements. The concentration of noble metals was, as expected, below the quantification limit, but recovery tests were successfully performed. Detection limits ranged from 0.02 to 0.1 μg L−1. Typical relative standard deviations were lower than 13% and the method was proven successful as an alternative to carry out the analysis of mildly diluted seawater samples.
- This article is part of the themed collections: Analytical Atomic Spectrometry in South America and JAAS 2018 Most Downloaded Articles


 Please wait while we load your content...
Please wait while we load your content...