Investigation of the iron oxidation state in ovarian cancer using synchrotron micro X-ray absorption near edge structure – preliminary results
Abstract
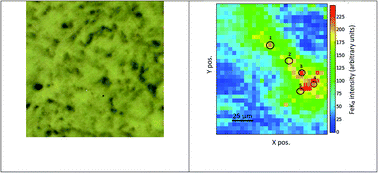
Iron as a trace element plays a role in regulating the homeostasis of the human body and therefore the concentration of iron in the body must be carefully monitored. Both deficiency and overload of iron could have a serious effect on health. Iron in excess can be carcinogenic in different ways. As a catalyst of Fenton's reaction it leads to the creation of reactive oxygen species which causes oxidative stress and DNA damage, among other things. Iron can also reduce the response of the immune system to cancer and induce increased cancer cell proliferation. Ovarian cancer is one of the most common neoplasms in women and has a low survival rate. For this reason, there is strong motivation to deepen the understanding of the processes that lead to the disease and take place in ovarian cancer tissue. In this study, cryo micro X-ray absorption near edge structure spectroscopy was used to analyse the oxidation state of iron in ovarian cancer samples. For calibration, organic and inorganic reference samples containing Fe in different oxidation states were used. To determine the trace element in the neoplasm samples, the pre-edge region, white line and absorption edge position of Fe K-edge XANES spectra were examined and compared to those of the recorded reference materials. A comparison of these XANES spectral features shows the existence of differences in the iron oxidation state between the neoplasm samples and suggests the possibility that a combination of iron ions can occur in one sample. Overall, further investigations are required to make a better comparison and determine what organic compounds are contained in the samples and possibly carry out a statistical analysis.
- This article is part of the themed collection: Atomic spectrometry for the analysis of biological samples


 Please wait while we load your content...
Please wait while we load your content...