The confined space electron transfer in phosphotungstate intercalated ZnAl-LDHs enhances its photocatalytic performance for oxidation/extraction desulfurization of model oil in air†
Abstract
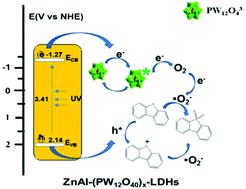
Deep desulfurization of fuels is a hot issue in the environmental protection field in recent years. Phosphotungstate intercalated hydrotalcites (ZnAl-(PW12O40)x-LDHs) with varying amounts of PW12O403− were prepared by an exfoliation-reassembly method, and their structures and properties were characterized by diverse techniques. The present process uses ZnAl-(PW12O40)x-LDHs as the catalyst, air as the oxidant, and acetonitrile as the extractant to remove dibenzothiophene (DBT) from model oil under UV irradiation. Results show that ZnAl-(PW12O40)0.07-LDHs has high photocatalytic activity at ambient temperature and pressure. 95.3% desulfurization can be achieved after 3 h UV irradiation at a catalyst dosage of 1 g L−1 and volume ratio of acetonitrile to oil = 1. There was no significant decline in catalyst performance after five catalytic cycles. Mechanism of photocatalytic oxidation desulfurization is revealed using methods, such as ESR, PL, electrochemical tests and active species trapping experiments. The main active species is found to be superoxygen anion free radicals (˙O2−) and photogenerated holes (h+). The high photocatalytic activity is mainly attributed to the synergistic effect between subject and object of the photocatalyst with confined space electron transfer, as well as extraction. A new system of photocatalytic oxidation/extraction desulfurization was constructed, and for the first time, LDHs material was applied to photocatalytic desulfurization of oil.


 Please wait while we load your content...
Please wait while we load your content...