Production of monosaccharides and whey protein from acid whey waste streams in the dairy industry†
Abstract
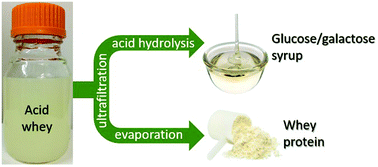
The kinetics of Brønsted-acid-catalyzed lactose hydrolysis in water and in acid whey solutions over sulfuric acid and solid acid catalysts were determined at temperatures from 120 to 160 °C. In water, lactose undergoes acid-catalyzed hydrolysis to produce glucose and galactose in near quantitative yield (activation energy 135.5 kJ mol−1). Additionally, lactose undergoes thermal (non-catalytic) hydrolysis to afford glucose and galactose monomers, while glucose and galactose undergo thermal reactions to form unaccountable degradation products (activation energies 156, 106, and 117 kJ mol−1, respectively). The rates at which the reactant and products undergo monomolecular and bimolecular acid-catalyzed degradation reactions are slow, and may be neglected for design purposes within the temperature range considered in this study. In acid whey solutions, hydrolysis of lactose affords 90% monosaccharide selectivity. Undesired reactions catalyzed by the proteins and non-protein nitrogen-containing species present in acid whey result in further degradation of the reactants and products. Ultrafiltration removes the protein and fat from the acid whey without removing lactose, but it does not remove the non-protein nitrogen species responsible for unwanted side reactions and catalyst deactivation, resulting in a 23% increase in hydrolysis rate but no change to the monosaccharide selectivity. Activated carbon treatment removes 33% of the non-protein nitrogen from ultrafiltration acid whey permeate and improves the monosaccharide selectivity to 93% but does not improve the rate, while nanofiltration produces a monosaccharide selectivity of 92% and increases the rate an additional 13% compared to ultrafiltration. Experiments comparing the solid acid catalyst Amberlyst 70 to mineral acid catalysts in aqueous lactose solutions at 150 °C show that both types of catalyst produce monosaccharide selectivities above 94% at high conversion (above 90%). Informed by these observations, we propose a process to convert acid whey waste streams into whey protein concentrates and glucose/galactose syrups, which would effect a significant reduction in the water, energy and carbon footprints associated with whey waste streams in the dairy industry.


 Please wait while we load your content...
Please wait while we load your content...