Development of CO2 phase change absorbents by means of the cosolvent effect†
Abstract
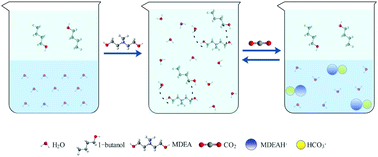
The CO2 phase change absorbent, CPCA, as a novel alternative absorbent, has attracted great attention recently. In this work, the CPCAs composed of MDEA/C4–C6 alcohol/H2O were developed by means of the cosolvent effect. The cosolvent effect of MDEA on the solubility of C4–C6 alcohols in H2O was verified. The CPCAs composed of MDEA/1-butanol/H2O were further examined including the species distributions, absorption rate, cycling capacity and regenerability. The initial absorption rates of all CPCAs were higher than that of 30% MDEA, and its maximum value was 2.5 times higher. A different behavior was shown for the CPCAs that an increase in the absorption rate was observed for a period of time when the CPCAs became turbid. The optimal CO2 cycling capacity of CPCA was 2.48 mol CO2 kg−1, which was 70% higher compared to 30 wt% MDEA.


 Please wait while we load your content...
Please wait while we load your content...