Mechanistic studies of base-catalysed lignin depolymerisation in dimethyl carbonate†
Abstract
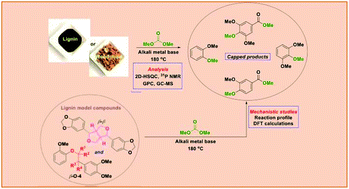
The depleting fossil reservoirs have stimulated global research initiatives on renewable lignin feedstocks as sustainable alternatives to petroleum-derived aromatics. Base-catalysed depolymerisation (BCD) is regarded as an economical and efficient approach for the valorisation of technical lignins. The major limiting factor encountered during this process is the re-condensation of the formed phenolic products, which results in lower monomer yields. To diminish these side reactions, we selected alkali earth metal catalysts in dimethyl carbonate (DMC) to produce methylated phenol derivatives as the final products. Herein, we demonstrate for the first time a base-promoted depolymerisation process affording low-molecular weight oils in high yields (52–67 wt%) wherein the employed bases are used in truly catalytic quantities (with catalyst loadings of around 5 mol%). The general applicability of this methodology was proved on four different lignin samples (1 Kraft, 3 organosolv) using caesium carbonate and lithium tert-butoxide as catalysts. The 2D NMR studies on the post-reaction lignin samples showed a similar degradation of the major lignin linkages for both bases. A difference in the reduction of phenolic moieties was revealed by quantitative 31P NMR analysis. Furthermore, GPC analysis demonstrated a significant shift towards lower mass fragments for the Cs2CO3-catalysed lignin degradation. A detailed GC-MS analysis for these samples identified a range of methoxy capped-monomeric degradation products. The scope of this reaction system was further expanded to lignocellulosic biomass such as milled beechwood chips, which notably showed similar product distributions. Based on the correlation of the experimental observations for extracted lignin samples and model compound studies, a mechanistic pathway for the Cs2CO3-catalysed system was suggested. DFT calculations provided reaction pathways for the observed cleavage products.
- This article is part of the themed collection: 2018 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...