Electrochemical production of syngas from CO2 captured in switchable polarity solvents†
Abstract
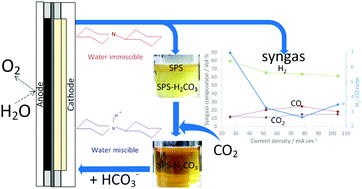
An operational advantage that enables the deployment of technologies for the valorization of CO2 can be achieved through the integration of the capture and conversion technologies. In that perspective, switchable polarity solvents (SPS) were assessed as re-usable capture-electrolyte media for the electrochemical production of syngas at low temperatures and pressures. A polymer electrolyte membrane cell was used to liberate captured CO2 gas in proximity to the cathode without the addition of CO2 gas. Due to the proximate location of release, the produced syngas had minimal CO2 dilution with H2 : CO ratios between 2 to 4. For the first time captured CO2 has been reduced to CO with conversions and yields over 70% and at current densities over 100 mA cm−2.
- This article is part of the themed collection: 2018 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...