Artificial sweetener saccharin disrupts intestinal epithelial cells’ barrier function in vitro
Abstract
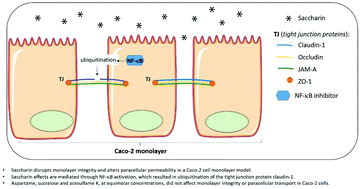
Scope: Consumption of non-nutritive sweeteners (NNS) is a dietary practice used by those who wish to lose weight or by patients on a sugar-restricted diet such as those with DM2. Although these substances are safe, possible biological interactions with the digestive tract, particularly in relation to intestinal permeability, have not been studied. Thus, the current work sought to investigate the action of different NNS on intestinal permeability using an in vitro Caco-2 cell model. Methods and results: Caco-2 cells were incubated with acesulfame K, aspartame, saccharin, or sucralose at equimolar concentrations. Acesulfame K, aspartame, and sucralose did not disrupt monolayer integrity in the cells. However, saccharin increased paracellular permeability and decreased transepithelial electrical resistance (TEER) via a non-cytotoxic mechanism. The levels of the tight junction protein claudin-1 were reduced in Caco-2 cells that had previously been exposed to saccharin. The inhibition of nuclear factor-κB (NF-κB) was able to prevent the reduction in TEER induced by saccharin treatment. Thalidomide, as an inhibitor of ubiquitin ligase, was able to prevent the decrease in claudin-1 protein expression and the TEER reduction in Caco-2 cells. Conclusions: Saccharin disrupts monolayer integrity and alters paracellular permeability in a Caco-2 cell monolayer model, via a mechanism involving NF-κB activation, resulting in the ubiquitination of the tight junction protein claudin-1. Saccharin consumption may potentially alter the intestinal integrity in humans.


 Please wait while we load your content...
Please wait while we load your content...