Curcumin attenuates doxorubicin-induced cardiotoxicity via suppressing oxidative stress and preventing mitochondrial dysfunction mediated by 14-3-3γ†
Abstract
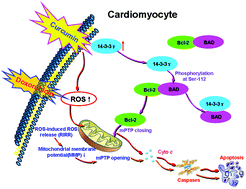
Doxorubicin (Dox) induces cardiotoxicity, thereby limiting its clinical application for chemotherapy of cancer. The mechanism of cardiotoxicity includes the production of excess intracellular ROS. 14-3-3s have been found to protect the myocardium against various types of injury. Curcumin (Cur) is a polyphenolic compound that is derived from turmeric and has multiple bioactivities, including anti-oxidative and radical-scavenging activities that exert cytoprotection. We hypothesize that the cardioprotective effects of Cur are exerted by regulating 14-3-3γ. To test the hypothesis, Dox-induced cardiotoxicity was used to establish an in vivo myocardial injury model in mice (in vivo) and primary cardiomyocytes (in intro). The effects of Cur were assessed by determining the heart rate and ECG's ST segments, as well as lactate dehydrogenase (LDH) and creatine kinase (CK) activities in the serum, caspase-3 activity, apoptosis rate, and histopathological changes of the myocardium (in vivo). In addition, cell viability, LDH, SOD, CAT, GPx, and caspase-3 activities, levels of ROS, MDA, and MMP, mPTP opening, and the apoptosis rate (in vitro) were evaluated. The expression of 14-3-3γ and Bcl-2 as well as the phosphorylation levels of Bad (S112) were determined by western blot analysis. Our results showed that Dox-induced injury to the myocardium was decreased by Cur treatment via upregulating the protein expression of 14-3-3γ in total protein and Bcl-2 expression on mitochondria, activating Bad (S112) phosphorylation, reducing the heart rate and ST segment, and reducing LDH and CK activities in the serum, thereby causing a reduction in caspase-3 activity, the apoptosis rate, and histopathological changes of the myocardium (in vivo). Furthermore, Dox treatment increased cell viability and MMP levels, decreased LDH and caspase-3 activity, ROS levels, mPTP opening, and the apoptosis rate (in vitro). However, the cardioprotective effects of Cur were attenuated by pAD/14-3-3γ-shRNA, an adenovirus that caused a knock-down of intracellular 14-3-3γ expression. In conclusion, this is the first study to demonstrate that Cur protected the myocardium against Dox-induced injury via upregulating 14-3-3γ expression, thereby promoting the translocation of Bcl-2 to mitochondria, suppressing oxidative stress, and improving mitochondrial function.


 Please wait while we load your content...
Please wait while we load your content...