Probing the selectivity of Li+ and Na+ cations on noradrenaline at the molecular level
Abstract
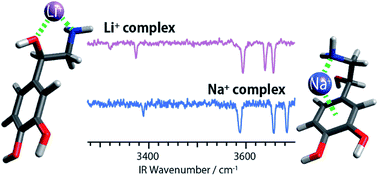
Although several mechanisms concerning the biological function of lithium salts, drugs having tranquilizing abilities, have been proposed so far, the key mechanism for its selectivity and subsequent interaction with neurotransmitters has not been established yet. We report ultraviolet (UV) and infrared (IR) spectra under ultra-cold conditions of Li+ and Na+ complexes of noradrenaline (NAd, norepinephrine), a neurotransmitter responsible for the body’s response to stress or danger, in an effort to provide a molecular level understanding of the conformational changes of NAd due to its interactions with these two cations. A detailed analysis of the IR spectra, aided by quantum chemical calculations, reveals that the Li+–noradrenaline (NAd–Li+) complex forms only an extended structure, while the NAd–Na+ and protonated (NAd–H+) complexes form both folded and extended structures. This conformational preference of the NAd–Li+ complex is further explained by considering specific conformational distributions in solution. Our results clearly discern the unique structural motifs that NAd adopts when interacting with Li+ compared with other abundant cations in the human body (Na+) and can form the basis of a molecular level understanding of the selectivity of Li+ in biological systems.
- This article is part of the themed collection: Advances in ion spectroscopy - from astrophysics to biology


 Please wait while we load your content...
Please wait while we load your content...
