Large amplitude motions within molecules trapped in solid parahydrogen†
Abstract
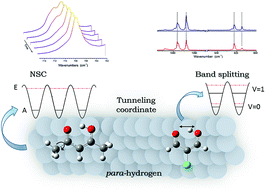
Molecules of the β-diketone and β-dialdehyde families were trapped in solid parahydrogen (pH2) to investigate the vibrational behavior of systems containing an intramolecular hydrogen bond (IHB). In the simplest β-diketone, acetylacetone (AcAc), H transfer related to the IHB is coupled with methyl torsions. In pH2, the study of nuclear spin conversion (NSC) in methyl groups allows the characterisation of the influence of these large amplitude motions on the vibrational modes. The deuteration of the OH group involved in the IHB has important consequences on the vibrational spectrum of the molecule and evidence of NSC in methyl groups is difficult to obtain. In the chlorine derivative (3-chloroacetylacetone), the H-transfer is no longer coupled with methyl torsion, and NSC has undetectable effects on the IR spectrum. A search of H tunnelling splitting in the IR spectra of β-dialdehydes trapped in pH2 was performed. A few modes of 2-chloromalonaldehyde appear as doublets and were assigned to tunnelling levels. The spectroscopic results related to large amplitude motions are detailed and discussed, highlighting puzzling effects.
- This article is part of the themed collection: Quantum effects in small molecular systems


 Please wait while we load your content...
Please wait while we load your content...