Quenching H2O2 residuals after UV/H2O2 oxidation using GAC in drinking water treatment†
Abstract
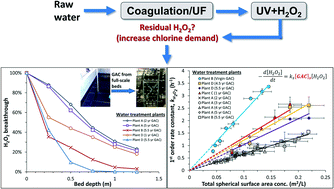
Pilot-scale and lab-scale experiments were performed to evaluate the ability of granular activated carbon (GAC) to quench hydrogen peroxide (H2O2). The GAC was harvested from 5 drinking water treatment plants and included two GACs that were specifically designed for this purpose (“catalytic”), and 3 that were used solely to adsorb taste and odour compounds (“normal”). All GACs indicated very little change in performance with time for GAC up to 9 years old (the oldest tested). Batch kinetic tests revealed that the catalytic GACs had a higher reaction rate with H2O2 than the normal GACs, but under typical conditions (e.g. 4+ minute EBCT) the difference may not be important in practice. The observed first order decomposition rate of H2O2 increased linearly with the spherical surface area of the GAC particles, providing evidence that effectiveness is governed by particle size, implying that smaller GAC would be more effective for quenching H2O2.


 Please wait while we load your content...
Please wait while we load your content...