Behavior of silver nanoparticles in wastewater: systematic investigation on the combined effects of surfactants and electrolytes in model systems†
Abstract
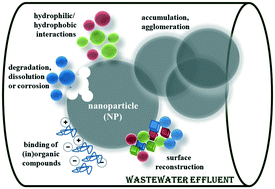
Due to the wide and increasing application of engineered silver nanoparticles (AgNPs) in commercial products, their release and disposal into aquatic environmental compartments is unavoidable. The final environmental fate of AgNPs depends on their stability, behavior and lifetime in a particular system. This study aimed to systematically investigate the aggregation and dissolution behavior of AgNPs in model aquatic systems including a range of different pH values, different concentrations of mono- and divalent electrolytes, and the presence of non-ionic, anionic and cationic surfactants used in commercial detergents. The investigation was performed by a combination of surface plasmon resonance (SPR) techniques, dynamic light scattering (DLS) and electrophoretic light scattering (ELS) methods, inductively coupled plasma mass spectroscopy (ICPMS), transmission electron microscopy (TEM), and atomic force microscopy (AFM). The obtained results highlighted the importance of pH, ionic strength and interaction with surfactants relevant to the laundry cycle to the stability and environmental fate of citrate-coated AgNPs. An important implication of this study is that the interaction of AgNPs with surfactants present in wastewater effluents could potentially become more environmentally hazardous due to the increased persistence and dissolution of AgNPs.


 Please wait while we load your content...
Please wait while we load your content...