Quasi single cobalt sites in nanopores for superior catalytic oxidation of organic pollutants†
Abstract
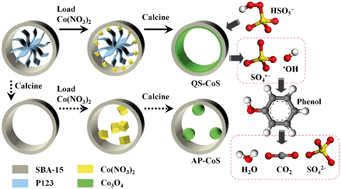
The reactivity of a supported metal catalyst is closely related to the dispersion degree of the active sites. The main objective of this study is to provide an effective strategy to promote the dispersion degree of cobalt sites on a porous material, i.e. SBA-15, and thus enhance the catalytic performances in advanced oxidation processes (AOPs). Herein, by directly employing the as-synthesized SBA-15 before the template removal as the support, for the first time, we develop a strategy to fabricate the quasi single cobalt sites in the naosized pores of SBA-15 (QS-CoS) with no particle aggregation. Our findings indicate that the confined space as well as abundant silicon hydroxyl groups in the as-synthesized SBA-15 is responsible for the formation of the resultant quasi single cobalt sites in the form of Co–O–Si. The QS-CoS catalysts are found to be highly efficient for peroxymonosulfate (PMS) activation to generate SO4˙− and ˙OH radicals. Under the dominant role of SO4˙−, 100% phenol degradation only takes 10 min on the 3.0QS-CoS sample. Furthermore, cobalt leaching is feasibly under control by regulating the reaction solution with a constant pH value of 7. In comparison, the aggregated Co3O4 particles appear on SBA-15 (AP-CoS) in the samples derived from calcined SBA-15 without the template. Up to 180 min, only 97.4% of phenol can be removed for the 3.0AP-CoS sample. Our synthesis strategy utilizing a confined space presents a wide range of possibilities for the further development of environmental or energy materials.


 Please wait while we load your content...
Please wait while we load your content...