Emerging investigator series: heterogeneous reactions of sulfur dioxide on mineral dust nanoparticles: from single component to mixed components†
Abstract
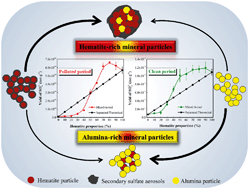
Sulfate compounds formed on mineral dust nanoparticles contribute significantly to severe haze events. Atmospheric particles are complex mixtures that comprise various species and little is known about the heterogeneous conversion of sulfur dioxide (SO2) on multi-composition nanoparticles. In this study, the heterogeneous reaction of SO2 on hematite–alumina mixtures is investigated using in situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS). Reactions in the presence of SO2 (polluted period) and its subsequent absence (clean period) are included. It is found that sulfite and sulfate species are the dominant products on alumina and hematite-containing mixtures, respectively. Since the uptake coefficients calculated using both Brunauer–Emmett–Teller (BET) surface area and geometric surface area present similar ‘S’ shape uptrends with increasing hematite proportion, a logistic regression model is employed for further predictions. Sulfate formation still occurs during the clean period, which can be attributed to the physical adsorption of SO2 during the polluted period. In both the periods, hematite-rich mixtures exhibit higher sulfate yields compared to their corresponding theoretical values based on the contribution of each individual component, whereas alumina-rich mixtures present an opposite trend, implying synergistic and antagonistic effects, respectively. A simplified mechanism is demonstrated and the environmental implications are discussed.
- This article is part of the themed collections: Best Papers 2018 – Environmental Science: Nano and Emerging Investigators Series


 Please wait while we load your content...
Please wait while we load your content...