Hydroxyl radical scavenging by cerium oxide nanoparticles improves Arabidopsis salinity tolerance by enhancing leaf mesophyll potassium retention†
Abstract
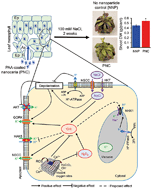
Salinity is a widespread environmental stress that severely limits crop yield worldwide. Cerium oxide nanoparticles (nanoceria) have the unique capability of catalytically reducing levels of stress-induced reactive oxygen species (ROS) including hydroxyl radicals (˙OH) that lack enzymatic scavenging pathways. The underlying mechanisms of how nanoceria ROS scavenging augments plant tolerance to environmental stress are not well understood. Herein, we demonstrate that catalytic ˙OH scavenging by nanoceria in Arabidopsis thaliana leaves significantly improves mesophyll K+ retention, a key trait associated with salinity stress tolerance. Leaves with mesophyll cells interfaced with 50 mg L−1 poly(acrylic acid) coated nanoceria (PNC) have significantly higher (P < 0.05) carbon assimilation rates (85%), quantum efficiency of photosystem II (9%), and chlorophyll content (14%) compared to controls after being exposed to 100 mM NaCl for 3 days. PNC infiltrated leaves (PNC-leaves) under salinity stress exhibit lower ROS levels – including hydroxyl radical (41%) and its precursor hydrogen peroxide (44%) – and one fold higher (P < 0.05) cytosolic K+ dye intensity in leaf mesophyll cells relative to controls. Non-invasive microelectrode ion flux electrophysiological (MIFE) measurements indicated that PNC-leaves have about three-fold lower NaCl-induced K+ efflux from leaf mesophyll cells compared to controls upon exposure to salinity stress. The ROS-activated nonselective cation channels (ROS-NSCC) in the plasma membrane of leaf mesophyll cells were identified as the main ˙OH-inducible K+ efflux channels. Long term catalytic scavenging of ˙OH in leaves by PNC enhances plant photosynthetic performance under salinity stress by enabling plasma membrane channels/transporters to coordinately retain higher levels of K+ in the leaf mesophyll cell cytosol. PNC augmented plant ROS scavenging provides a key tool for understanding and improving plant tolerance against abiotic stresses such as salinity.
- This article is part of the themed collection: Best Papers 2018 – Environmental Science: Nano


 Please wait while we load your content...
Please wait while we load your content...
